What is Immunity?
Immunity is a complex process with both innate and adaptive immunity. The adaptive arm of the immune system comprises both antibody responses and T cell responses. Most recently, the role of T cells and antibodies has become an important point of discussion about which confers the most effective immunity, especially in relation to COVID-19 infection and vaccination.¹ Immunity and clearance of a viral infection is carried out by the T cells and antibodies. T cells can kill infected cells after recognising parts of the virus presented by the MHC complex, whilst antibodies can bind to the virus in the nasal mucosa before a cell becomes infected thus providing protection. The immunology community is divided over whether true immunity comes from T cells and some believe only a truly sterilising serum contains antibodies.²
Correlation between antibody concentration and T cell concentration:
The correlation between antibody concentration and T cell concentration is ~50% (r= 0.5) from a number of studies¹. Antibody levels have been reported by others as correlates of immunity³ in 18 vaccines⁴. The message about vaccination during the pandemic has evolved, from the initial objective to prevent infection to using vaccines to prevent serious infection and hospitalisation. Whilst T cells and antibodies wane ¹ ⁵ over time, the rapid production of both following priming by vaccination prevents serious infection.
Can antibodies be used as a correlate for SAR-CoV-2 ?
Investigations of antibodies as a correlate of immunity to SARS-CoV-2 have been mixed; some report unsuccessful correlations⁶ others report robust correlation⁷. Similar investigations correlating antibody levels and efficacy have previously been explored for 18 vaccines ⁸ ⁹ with some successes. Elsewhere, potential correlates of protection have been investigated across the complex immune response triggered by infection and other vaccines ¹⁰, most critically B cells, T cells and antibody levels.
The Attomarker COVID-19 Antibody Immunity Test is a fully quantitative immunoassay calibrated against the conserved S2 regions of the spike protein, omicron spike protein, nucleocapsid and receptor binding domain proteins with recombinant antibodies calibrated against the mass standard material NIST Human Antibody. ¹¹ ¹²
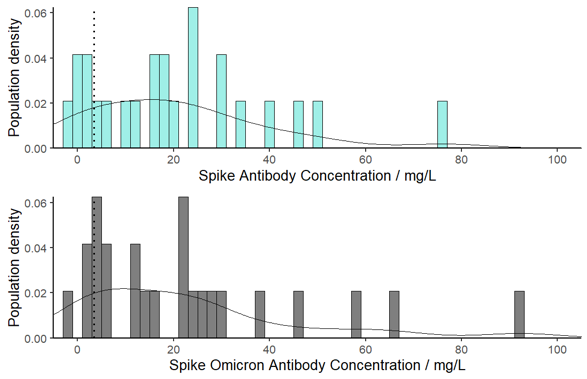
Immune Responses in the Population
Quantitative measurement of antibody levels can be used to profile the immune response in the population. There is a significant spectrum of response in the community derived from different combinations of vaccines interspersed with natural infections:
- AstraZeneca, AstraZeneca, Pfizer
- Pfizer, Pfizer, Pfizer
- AstraZeneca, AstraZeneca, AstraZeneca, Moderna
The current observed range of antibody levels is from 0.1 ng/L to 190 mg/L; a similar range is observed for antibody levels to the Omicron spike protein, being typically 80% of the Wuhan vaccine response. Approximately 1/3 of the population fall below the protective nasal threshold 100-days after triple vaccination. Public Health England report the triple-jab boosted vaccine is 60 -70% effective against preventing infection with Omicron and falling to 30-40% effective after 15 weeks ¹³, consistent with the 60-day half-life.
Conclusions:
- COVID-19 antibody immunity has been tested against T cells and there is a quantitative correlation which is reassuring for such a complex process as adaptive immunity.
- Antibody immunity can be readily measured and can be used for population profiling.
Contact: Prof Andrew Shaw, Andrew.Shaw@attomarker.com
References:
- Jiang X-L, Wang G-L, Zhao X-N, et al. Lasting antibody and T cell responses to SARS-CoV-2 in COVID-19 patients three months after infection. Nature Communications 2021; 12(1): 897.
- Khoury DS, Cromer D, Reynaldi A, et al. Neutralizing antibody levels are highly predictive of immune protection from symptomatic SARS-CoV-2 infection. Nature Medicine 2021; 27(7): 1205-11.
- Feng S, Phillips DJ, White T, et al. Correlates of protection against symptomatic and asymptomatic SARS-CoV-2 infection. medRxiv 2021: 2021.06.21.21258528.
- Plotkin SA. Correlates of protection induced by vaccination. Clin Vaccine Immunol 2010; 17(7): 1055-65.
- Goldberg Y, Mandel M, Bar-On YM, et al. Waning Immunity after the BNT162b2 Vaccine in Israel. The New England journal of medicine 2021.
- Abbasi J. The Flawed Science of Antibody Testing for SARS-CoV-2 Immunity. Jama 2021; 326(18): 1781-2.
- Earle KA, Ambrosino DM, Fiore-Gartland A, et al. Evidence for antibody as a protective correlate for COVID-19 vaccines. Vaccine 2021; 39(32): 4423-8.
- Thakur A, Pedersen LE, Jungersen G. Immune markers and correlates of protection for vaccine induced immune responses. Vaccine 2012; 30(33): 4907-20.
- Plotkin SA. Correlates of protection induced by vaccination. Clin Vaccine Immunol 2010; 17(7): 1055-65.
- Koch T, Mellinghoff SC, Shamsrizi P, Addo MM, Dahlke C. Correlates of Vaccine-Induced Protection against SARS-CoV-2. Vaccines 2021; 9(3).
- James-Pemberton PH, Helliwell MW, Olkhov RV, et al. Fully Quantitative Measurements of the Antibody Levels for SARS-CoV-2 Infections and Vaccinations calibrated against the NISTmAb Standard IgG Antibody. medRxiv 2022: 2022.07.12.22277533.
- James-Pemberton PH, Helliwell MW, Olkhov RV, et al. Vaccine, Booster and Natural Antibody Binding to SARS-CoV-2 Omicron (BA.1) Spike Protein and Vaccine Efficacy. medRxiv 2022: 2022.07.12.22277539.
- UKHSA. How well do vaccines protect against Omicron? What the data shows. 2022. https://ukhsa.blog.gov.uk/2022/02/10/how-well-do-vaccines-protect-against-omicron-what-the-data-shows/ (accessed 14/03/2022)
